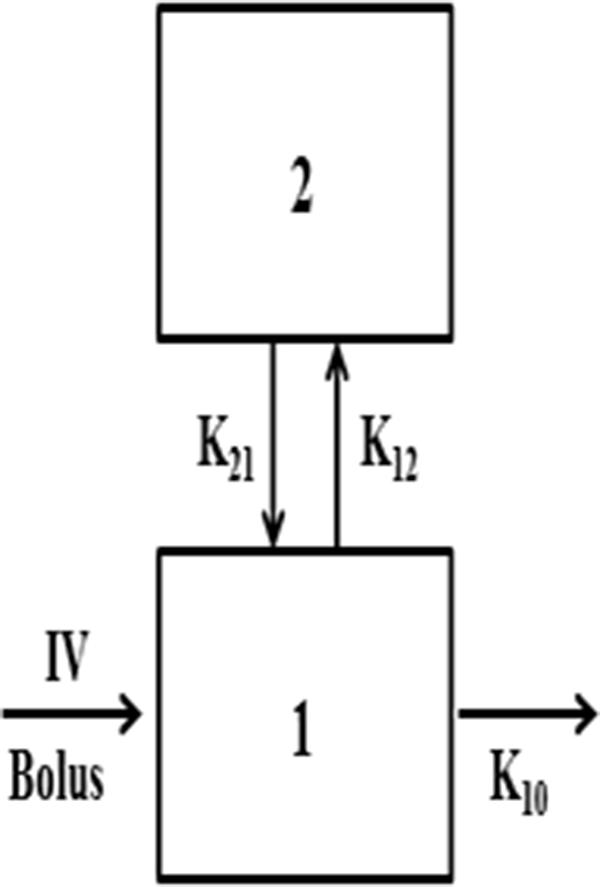
12 Further, the manufacturer of VEGF-Trap, Regeneron Pharmaceuticals Inc. In one PK study, the intravitreal half-life of VEGF-Trap was found to be 4.58 days, as assessed by positron emission tomography/computed tomography imaging. 11 Accordingly, it is hypothesized that VEGF-Trap will require less-frequent administration by intravitreal injection compared with ranibizumab and bevacizumab, although intraocular pharmacokinetic (PK) studies for VEGF-Trap are lacking. 6, 7, 8, 9, 10 VEGF-Trap has greater binding affinity for VEGF than ranibizumab and bevacizumab, 6 suggesting that it may have a substantially longer duration of action in the eye, as predicted by a mathematical model. The novel anti-VEGF agent VEGF-Trap was developed for the treatment of VEGF-associated intraocular conditions, including exudative AMD and macular edema secondary to RVO and diabetic retinopathy. 1, 2, 3, 4 In addition, intravitreal administration of bevacizumab (Avastin Genentech Inc.) has been used to treat exudative AMD and other VEGF-associated conditions, although this drug is only approved for the systemic treatment of metastatic colon cancer. Intravitreal ranibizumab injection (Lucentis Genentech Inc., San Francisco, CA, USA), approved by the United States Food and Drug Administration for the treatment of exudative AMD 1, 2 and macular edema subsequent to RVO, 3 has been the first-line treatment for diseases affecting sight, and has shown notable efficacy in vision improvement. The concentration of VEGF-Trap peaked at 1 h after injections in all eye tissues studied.Īnti-vascular endothelial growth factor (anti-VEGF) agents have been widely used in numerous diseases affecting sight, including exudative age-related macular degeneration (AMD), macular edema subsequent to retinal vein occlusion (RVO) or diabetic retinopathy, and neovascular glaucoma. This is shorter than that of bevacizumab (6.99 days) and longer than that of ranibizumab (2.51 days), as shown in studies using the same experimental settings. The vitreous half-life of VEGF-Trap is 3.63 days.
Total exposure of the aqueous humor and retina/choroid to VEGF-Trap was 39.4% and 11.9% of vitreous exposure, respectively. Area under the curve from time 0 to the end point was 10009.8, 3945.1, and 1189.3, respectively. Estimated half-life of VEGF-Trap in vitreous, aqueous humor, and retinal/choroid was 87.1, 36.8, and 35.0 h, respectively, and estimated mean residence time was 125.7, 53.1, and 50.5 h, respectively. A one-compartment model of distribution was selected as the final model for all tissues studied. Maximum concentration of VEGF-Trap was achieved at 1 h in all three tissues. Concentration of VEGF-Trap in vitreous, aqueous humor, and retina/choroid was determined using an indirect enzyme-linked immunosorbent assay and analyzed to obtain pharmacokinetic properties. Eyes were enucleated 1 h and 1, 2, 5, 14, and 30 days after injections and immediately frozen at −80 ☌. VEGF-Trap was intravitreally injected in 18 rabbit eyes. To determine intraocular pharmacokinetic properties of intravitreally injected vascular endothelial growth factor (VEGF)-Trap in a rabbit model.
